The agency approved Gilead Sciences'
pill Truvada as a preventive measure for people who are at high risk of
acquiring HIV through sexual activity, such as those who have
HIV-infected partners.
Public health advocates
say the approval could help slow the spread of HIV, which has held
steady at about 50,000 new infections per year for the past 15 years.
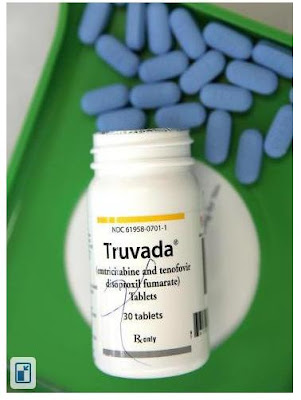 |
| Truvada can prevent people from contracting HIV when used as a precautionary measure |
Gilead Sciences has marketed Truvada, which can
cost up to $14,000 a year, since 2004 as a treatment for people who are
already infected with the virus.
But starting
in 2010, studies showed that the drug could actually prevent people from
contracting HIV when used as a precautionary measure. A three-year
study found that daily doses cut the risk of infection in healthy gay
and bisexual men by 42%, when accompanied by condoms and counseling.
Last
year, another study found that Truvada reduced infection by 75% in
heterosexual couples in which one partner was infected with HIV and the
other was not.
Because Truvada is on the market to manage HIV, some doctors already prescribe it as a preventive measure. FDA approval will allow Gilead Sciences to market the drug for that use, which could dramatically increase prescribing.
STORY: Truvada Q and A

No comments:
Post a Comment